About Citric Acid Properties
Citric acid (C₆H₈O₇) is a versatile organic acid with unique physical and chemical properties that make it indispensable across multiple industries.
Molecular Structure
Tricarboxylic acid with three carboxyl groups
Crystalline Form
White crystalline powder or colorless crystals
High Solubility
Highly soluble in water and alcohol
Acidic Nature
Strong acidic taste with pH ~2.2 (1% solution)
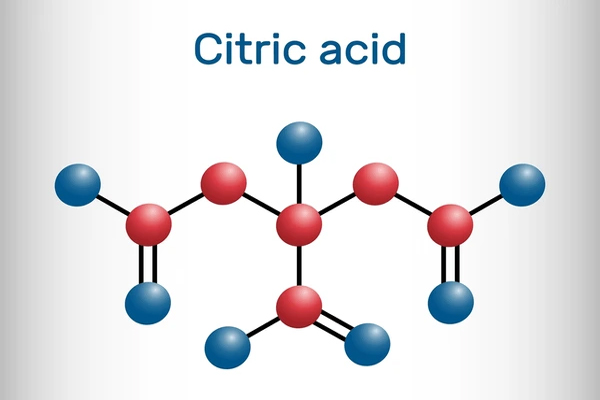
Crystalline structure of pure citric acid
Physical Properties
| Physical Characteristics | |
|---|---|
| Appearance | White crystalline powder or colorless translucent crystals |
| Odor & Taste | Odorless with strongly acidic taste |
| Specific Gravity | 1.542 g/cm³ at 20°C |
| Melting Point | 153°C (Anhydrous) | 100°C (Monohydrate, with decomposition) |
| Boiling Point | Decomposes above 175°C |
| Bulk Density | 0.9 - 1.0 g/cm³ (powder) |
| Refractive Index | 1.493 - 1.509 |
Physical characteristics of citric acid
Key Physical Properties
- Hygroscopic: Monohydrate form absorbs moisture from air
- Efflorescent: Loses water of crystallization in dry air
- Stable: Stable under normal storage conditions
- Non-volatile: Does not evaporate at room temperature
Chemical Properties
| Chemical Characteristics | |
|---|---|
| Chemical Formula | C₆H₈O₇ (Anhydrous) | C₆H₈O₇·H₂O (Monohydrate) |
| Molecular Weight | 192.12 g/mol (Anhydrous) | 210.14 g/mol (Monohydrate) |
| CAS Number | 77-92-9 (Anhydrous) | 5949-29-1 (Monohydrate) |
| EINECS Number | 201-069-1 |
| Acidity (pKa) | pKa₁ = 3.13 | pKa₂ = 4.76 | pKa₃ = 6.40 |
| pH (1% solution) | Approximately 2.2 |
| Heat of Combustion | 1980 kJ/mol |
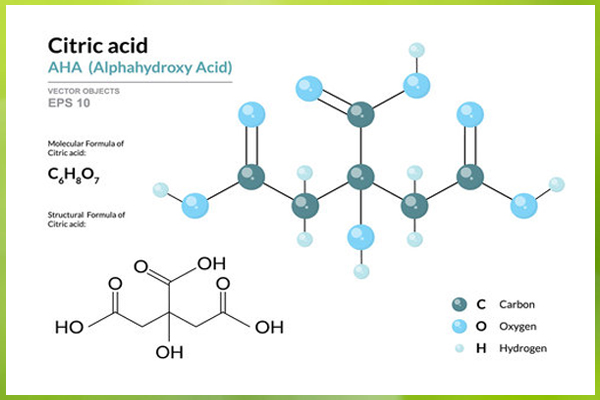
Chemical structure of citric acid
Chemical Behavior
- Weak Organic Acid: Tribasic acid with three carboxyl groups
- Complex Formation: Strong chelating agent for metal ions
- Thermal Stability: Decomposes above 175°C to aconitic acid
- Biodegradable: Completely biodegradable under aerobic conditions
Solubility Properties
Water Solubility vs Temperature
| Temperature (°C) | Solubility (g/100g H₂O) | Crystal Form |
|---|---|---|
| 0 | 41.0 | Monohydrate |
| 10 | 54.0 | Monohydrate |
| 20 | 59.2 | Monohydrate |
| 30 | 64.3 | Monohydrate |
| 40 | 68.6 | Anhydrous |
| 50 | 70.9 | Anhydrous |
| 60 | 73.5 | Anhydrous |
| 70 | 76.2 | Anhydrous |
| 80 | 78.8 | Anhydrous |
| 90 | 81.4 | Anhydrous |
| 100 | 84.0 | Anhydrous |
Organic Solvent Solubility (25°C)
| Solvent | Solubility (g/100g) | Remarks |
|---|---|---|
| Water | 59.2 | Highly soluble |
| Ethanol | 62.8 | Freely soluble |
| Methanol | 197.0 | Very soluble |
| Ethyl Acetate | 5.28 | Soluble |
| Acetone | 1.52 | Slightly soluble |
| Diethyl Ether | 2.17 | Slightly soluble |
| Chloroform | 0.007 | Practically insoluble |
Specification Comparison: Monohydrate vs Anhydrous
| Parameter | Specification | Citric Acid Monohydrate | Citric Acid Anhydrous |
|---|---|---|---|
| Identity & Composition | |||
| Chemical Formula | - | C₆H₈O₇·H₂O | C₆H₈O₇ |
| Molecular Weight | - | 210.14 g/mol | 192.12 g/mol |
| Assay (Titrimetric) | 99.5% - 100.5% | 99.5% min | 99.5% min |
| Physical Properties | |||
| Appearance | White crystalline | ✓ Complies | ✓ Complies |
| Water Content | - | 7.5% - 9.0% | ≤ 0.5% |
| Sulphated Ash | ≤ 0.1% | ≤ 0.05% | ≤ 0.05% |
| Purity Tests | |||
| Heavy Metals (as Pb) | ≤ 10 ppm | ≤ 10 ppm | ≤ 10 ppm |
| Oxalic Acid | ≤ 350 ppm | ≤ 100 ppm | ≤ 100 ppm |
| Sulfate (SO₄) | ≤ 150 ppm | ≤ 150 ppm | ≤ 150 ppm |
| Arsenic (As) | ≤ 1 ppm | ≤ 1 ppm | ≤ 1 ppm |
| Iron (Fe) | ≤ 50 ppm | ≤ 50 ppm | ≤ 50 ppm |
| Chloride (Cl) | ≤ 50 ppm | ≤ 50 ppm | ≤ 50 ppm |
| Storage & Stability | |||
| Hygroscopicity | - | Hygroscopic | Non-hygroscopic |
| Storage Temperature | - | Below 25°C | Below 30°C |
| Shelf Life | - | 36 months | 48 months |
Monohydrate Form
Contains one water molecule per citric acid molecule. Ideal for humid environments and applications where moisture content is not critical.
Anhydrous Form
Completely water-free. Preferred for moisture-sensitive applications and products requiring precise water content control.
Form Selection Guide
Understanding when to use Monohydrate vs Anhydrous citric acid is crucial for optimal performance in your application:
Choose Monohydrate When:
- Cost-effectiveness is priority
- Application is not moisture-sensitive
- Processing in humid conditions
- Food and beverage applications
Choose Anhydrous When:
- Precise water content control needed
- Moisture-sensitive applications
- Long-term storage requirements

Both Forms Available
IMLI supplies both monohydrate and anhydrous forms in various food grades