Overview
Official specification and test procedures for Citric Acid (Anhydrous/Monohydrate) as per pharmacopoeial standards.
- Molecular Formula C₆H₈O₇
- Molecular Weight 192.12 g/mol
- Melting Point 153°C
- Test Methods 16 Parameters
- Assay Range 99.5-100%
- Basis Anhydrous

IMLI Quality Control Laboratory
Physical Properties
A white, crystalline powder, colorless crystals or granules.

Citric Acid Crystalline Powder
Very soluble in water, freely soluble in alcohol, sparingly soluble in ether.
Water
Very SolubleAlcohol
Freely SolubleEther
Sparingly SolubleIdentification Tests
| Test | Method | Observation |
|---|---|---|
| A | Dissolve 1 g in 10 ml water | Strongly acidic |
| B | IR Spectrophotometry | Match reference spectrum |
| C | Acetic anhydride + Pyridine | Red color develops |
| D | Calcium chloride test | White precipitate |
| E | Water content test | Complies with limit |
Color & Clarity Tests
4 Appearance of Solution
Test Solution: Dissolve 2.0 g in water and dilute to 10 ml.
Reference Solutions: Prepare BY7 and Y7 solutions as per pharmacopoeia.
Examine in diffused daylight against white background.
5 Readily Carbonizable Substances
Test: Heat 1.0 g with 10 ml H₂SO₄ at 90°C for 60 min.
Reference: Mix 1 ml red + 9 ml yellow primary solution.
Inorganic Impurities
| Parameter | Limit | Method Reference |
|---|---|---|
| 6. Oxalic Acid | ≤ 350 ppm | |
| 7. Sulfate | ≤ 150 ppm | |
| 8. Arsenic | ≤ 1 ppm | |
| 9. Barium | Passes test | |
| 10. Calcium | ≤ 200 ppm | |
| 11. Iron | ≤ 50 ppm | |
| 12. Chloride | ≤ 50 ppm |
Heavy Metals & Residue
13 Heavy Metals
Sample Solution: Dissolve 5.0 g in dilute NaOH, dilute to 50 ml.
Standard Solution: 10 ml of Lead standard (1 ppm Pb).
Procedure: Add acetate buffer pH 3.5 and thioacetamide reagent, allow standing for 2 minutes.
Acceptance: Test solution color ≤ Standard solution color.
14 Sulphated Ash
% Ash = [(W₃ - W₁) / (W₂ - W₁)] × 100
15 Water (Karl Fischer)
Karl Fischer Titration
Assay (Purity)
16 Titrimetric Assay
1 ml of 1M NaOH ≡ 64.03 mg of C₆H₈O₇
% Assay = (B.R. × M × 64.03 × 100 × 100) / (0.1 × W × (100 - LOD))
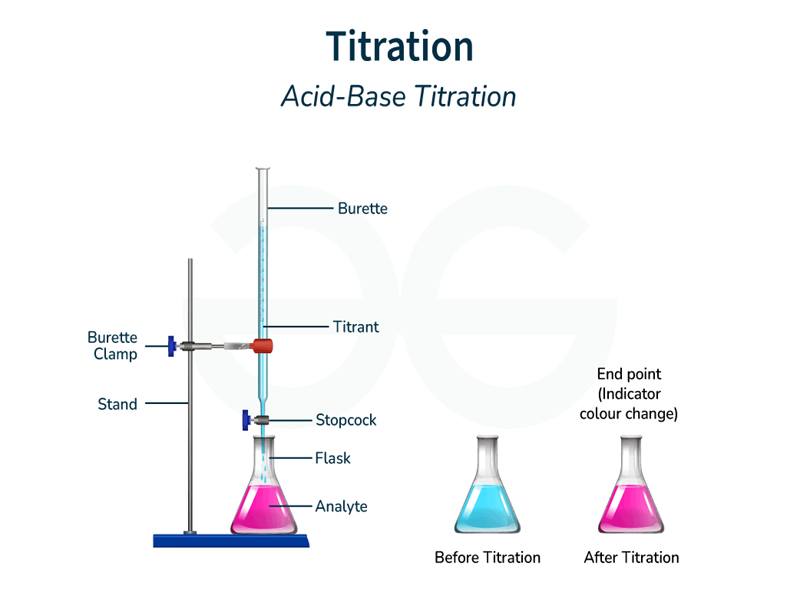
Titration Analysis
Procedure: Dissolve 0.550 g in 50 ml water. Titrate with 1M sodium hydroxide using phenolphthalein indicator.
Calculation:
% Assay = (Burette Reading × Actual Molarity × 64.03 × 100 × 100)
-----------------------------------------------------
(0.1 × Sample Weight (g) × (100 - %LOD))